-
The prognosis of Hepatitis B and C virus (HBV, HCV) infected patients is influenced by multiple factors, among which viral mutations and its nature of quasispecies can be closely related to all aspects of the disease progress of these patients, including chroni-calization, acute flares of chronic hepatitis, and the emerging of severe hepatitis as well as cirrhosis and hepatocellular carcinoma (2, 3, 4, 5).
It has been well stated that in any single host, HBV and HCV exist as closely related yet genetically distinguished variants known as quasispecies, based on the lack of proofreading of their polymerases (6). Variants possess the highest fitness to environment may exist as predominant quasispecies while others remain as minor variants. The proportion of these variants may fluctuate according to environmental changes so that to keep the whole population fit best to the changing circumstances. By these means, the quasispecies nature of HBV and HCV has endowed these hypervariable viruses competences of recrui-ting numerous variants to compete with any environ-mental pressures the population might encounter with. By sacrificing non-vital variants, reserving less-fit yet potential variants, and dominating one or several highest fit variant (s), a reservoir of various variants was generated which may possess a strong com-petence of adapting to and evolving with the changing environments (7, 11). Therefore, when facing with host immune pressure or antiviral drugs, potential fitful variants which may previously be minor quasi-species can be selected and accumulate to be dominant quasispecies. The quasispecies nature may be one of the main reasons for immune escape and drug resistance of these hypervariable viruses.
HTML
-
Antiviral therapy is one of the hottest issues for physicians dealing with chronic hepatitis B and C patients. For those chronically infected with hepatitis B, nucleoside analogs (NA) were considered effective in reducing viremia, convenient for oral admini-stration and less possible for the occurrence of side effects. However, the emerging drug resistance became the crucial bottleneck for the better appli-cation of this kind of reagents. The resistance rate to lamivudine (LAM) was reported to be 14% after one year's therapy, whereas this rate rose to 38%, 49%, 66% and 69% after 2, 3, 4 and 5 years respectively (9). After detection of the YMDD variant, it has once been considered that HBV develops drug-resistant mu-tations under the pressure of LAM (1). However, this recognition was adjusted to what is now widely accepted that this YMDD variant was one of the various previously existing minor quasispecies which 'happened' to be selected out and became dominant variant under the pressure of LAM (14).
With the development of adefovir (ADV), entecavir (ETV), telbivudine (L-dT) and pegylated interferon alpha (PEG-IFN-α), more choices are available for anti-HBV therapy at present and in the future, as a series of nucleoside analogs such as emtricitatine (FTC) and clevudine (L-FMAU) still coming (10). ETV has the highest competence of HBV inhibition, while both ADV and ETV have been shown to have much lower resistance rates compared to LAM. However, mutations against these new drugs still emerged (10). Meanwhile, physicians are now facing with new clinical questions such as: ⅰ. Primary selection of NA for NA-na ve patients, ⅱ. Prevention and management of NA resistance, ⅲ. Feasibility and strategy of combination or sequential NA therapy, and ⅳ. The appropriate occasion to discontinue NA therapy, etc. Undoubtedly, NA resistance is the key to these questions. Studies designed to investigate the longitudinal evolution of HBV quasispecies population under drug pressure were extremely helpful in under-standing the mechanism of NA resistance. It has been stated that long-term LAM therapy was responsible to the accumulation of LAM resistant variants, and closely related to virologic and biochemical break-through and the failure of antiviral therapy (14), whereas sequential nucleoside analog therapy may lead to emergence of multi-drug resistant HBV (Fig. 1) (19). A much recent study conducted in our lab showed complicated evolution of LAM and ETV resistant quasispecies variants during a four-year LAM-ETV sequential therapy. The results of these studies strongly indicated that individual treatment optimization will require sensitive methods capable of detecting the emergence of minor drug resistant variants before they acquire optimal replicative capacities and become dominance, while De novo combination therapy or initiation of drugs with higher competence of viral inhibition and lower resistance rates may prevent the emergence of multi-drug resistant mutants.
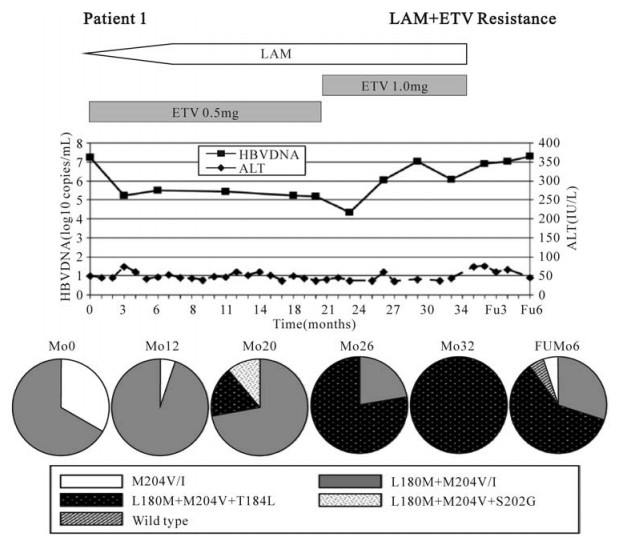
Figure 1. Clinical course and evolution of antiviral resistant mutations in patient 1, who had lamivudine-resistant HBV, and then was treated with combination of lamivudine and entecavir. HBV DNA levels in solid line and ALT levels in dashed line are plotted against time in months from the start of lamivudine and entecavir combination treatment. Proportions of clones at each time with various patterns of mutations are depicted as pie charts. Antiviral therapies are shown above each graph. HBV, hepatitis B virus; ALT, alanine aminotransferase; LAM, lamivudine; ETV, entecavir; Mo, month; FU, follow-up (12).
However, early detection of accumulating minor quasispecies is the bottleneck for current implemen-tation of this clinical test. The method of PCR-cloning-sequencing can have an accuracy of 3.33% by sequencing 30 clones for each sample, yet clinical application asks for more economic and convenient methods. Real-time PCR as well as gene chip technology may give technique support to this issue in the future.
-
Viral mutation is closely related to immune escape and tolerance. According to previous findings, the more complex the quasispecies composition is, the less possible that host clears the virus. Accordingly, more clones of HBV specific CTL can mean a much higher possibility of viral clearance (15, 16, 17). Long-term investigation of viral evolution in peri-natally HCV infected children indicated that the evolution of HCV quasispecies correlates strongly with hepatic injury, that biochemical evidence of hepatic injury was invariably associated with a mono-or oligoclonal viral population, whereas mild or no liver damage correlated with the early emergence of a heterogeneous viral quasispecies (Fig. 2) (8). There-fore, whether the host can eliminate the infected virus depends on both the virus factors and the host factors.

Figure 2. Long-term clinical course and evolution of the HCV quasispecies during long-term follow-up of perinatally acquired HCV infection in three representative children, two with high ALT levels (Patients 1 and 6), one of whom had an unusually severe hepatitis, with an ALT peak of 1, 213 IU_liter (Patient 6), and one with low ALT levels (Patient 8). (A, C, and E) Long-term clinical course of hepatitis C. The light blue areas denote ALT levels. The red horizontal bars denote positive assays for serum HCV RNA by PCR. The red lines denote the titer of serum HCV RNA on a logarithmic scale. The yellow horizontal bars denote the infant's nascent antibody response to HCV, as detected by third generation RIBA. (B, D, and F) Long-term evolution of the HCV quasispecies. Number of viral variants and genetic diversity (genetic distance among variants) of the HCV quasispecies within the 27 amino acids of the HVR1 were demonstrated. The vertical bars indicate the number and the proportion of viral variants within each sample. The dominant viral variant found in each patient at the first time point is indicated in blue; other variants are indicated by additional colors. Within the vertical bars, each variant is identified by a different color. The same color indicates identity between viral variants detected at different time points within each patient and not between different patients. The viral population diversity (black line) was calculated by mean Hamming distance from the predicted amino acid sequences obtained from each sample (16).
The activation of host immune system is a complicated process, which may be influenced by multiple factors. The evolving environmental pressures might lead to intricate adaptation of the virus quasispecies population, which can mean that viral strain which could be initially recognized by host immune system and lead to host immune activation can by some means be kept as minor quasispecies during the long run, and may again accumulate and become dominant quasispecies under certain circum-stances. It has been stated that secretory HBeAg can theoretically alleviate the immune attack initiated by CTLs (12, 18), while the A83 mutation in preC gene can lead to the intermittence of the synthesis of secretory HBeAg, and then lead to an activation of host immune system, and eventually result in hepatocellular injury, acute exacerbation of hepatitis and even severe or fulminant hepatitis (13). As the correlation between mutations in different genes of HBV genome is still poorly studied, it is quite possible that certain environmental driven mutations might be correlated with the known or unknown immune activation mutations. When the previous mutations were driven to be the dominant quasispecies, the latter will then involuntarily be dominant too and cause host immune activation.
The quasispecies nature of highly clinical related hypervariable viruses such as HCV, HIV and HBV is the upgraded definition of viral mutation from the population and evolution point of view. Further study on the mechanism of the evolution of viral quasis-pecies under certain environmental pressures and its interaction with host as well as novel techniques for the detection and analysis of quasispecies are urgently needed.













 DownLoad:
DownLoad: