HTML
-
Porcine reproductive and respiratory syndrome (PRRS) is characterized by reproductive failure in sows and gilts, along with increased preweaning mortality in piglets. The causative agent, PRRS virus (PRRSV), belongs to the family Arteriviridae, order Nidovirales (Guo et al. 2013). Immunologically, PRRSV infection is characterized by delayed humoral and cell-mediated responses (Chand 2012). PRRSV non-neutralizing antibody can be detected at 7 days post-infection, and this IgG Ab is primarily directed towards nucleocapsid (N) protein and provides no protection against PRRSV infection (Sun et al. 2008a, b). PRRSV-specific neutralizing antibody can be detected 3 weeks after infection. In the presence of neutralizing antibody, low levels of PRRSV replication still occur in lung and lymphoid tissues, indicating that other immune mechanisms could be involved in the complete elimination of the virus (Zhang et al. 2012). Cell-mediated immunity (CMI) can be detected 4 weeks post-infection. CMI plays an important role in the resolution of many viral infections through the function of cytotoxic T lymphocytes (CTLs) and T helper (Th) lymphocytes (Sun et al. 2008a, b). IFNγ is a pleiotropic cytokine produced by activated T cells and natural killer cells and plays a pivotal role in immune response (Lunney et al. 2010). IFNγ is the principal activator of macrophage function, modulates the growth and differentiation of cytotoxic T cells, activates NK cells, and regulates B cell Ig isotype switching to shift the balance towards a Th1-type response. As the major mediator of CMI response, IFNγ is associated with protection against PRRSV infection.
Porcine T cells can be divided into a number of subpopulations, including a prominent fraction of T cells expressing T cell receptors (TCR) with γδ chains (Gerner 2009). TCR-αβ T cells can be divided into four different subsets based on CD4 and CD8 expression, namely CD4+ T-helper cells, CD8+ CTLs, CD4+CD8+ Th/memory cells, and a fraction of CD4-CD8- cells. The existence of a considerable fraction of CD4+CD8+ T cells is a peculiarity of the porcine immune system. Th/memory cells are a major IFNγ-secreting cell population and possess memory, T-helper, and cytolytic properties (Sinkora and Butler 2009). Swine also possess a large percentage of γδ T lymphocytes in the peripheral circulation, which are capable of responding to various pathogens in both innate and specific immune responses (Murtaugh and Genzow 2011). Similar to αβ lymphocytes, γδ lymphocytes secrete cytokines, including IFNγ, proliferate, and exert antigendriven cytolytic activity (Kimman et al. 2009).
Several porcine lymphocyte subsets including T-helper, CTLs, Th/memory, and γδ T lymphocytes contribute to IFNγ-producing during PRRSV infection (Xiao et al. 2004). T cell secretion of IFNγ in response to PRRSV appears to be restricted to CD4+CD8+ Th/memory cells and CD4+ T-helper cells (Rodriguez-Carreno et al. 2002). However, the proportions and phenotypes of these IFNγ-secreting lymphocytes have not been explored. In this study, pigs were vaccinated with PRRSV-modified live vaccine (PRRSV-MLV) and then challenged with homologous VR2332 or heterologous JXA1 PRRS viruses to analyze the phenotypic characteristics and proportions of lymphocyte subsets contributing to IFNγ-producing.
-
Marc-145 cells were maintained in Modified Eagle's medium supplemented with 7% fetal bovine serum (FBS) containing 100 U penicillin/mL and 100 μg streptomycin/mL at 37 ℃ with 5% CO2. Virus stocks were prepared and titered in Marc-145 cells and stored in aliquots at -80 ℃ until use. For virus infection and titration, minimum Eagle's medium (MEM) supplemented with 2% FBS was used. PRRSVmodified live virus vaccine (MLV) was bought from Boehringer Ingelheim Vetmedica Inc. PRRSV VR-2332 and JXA1 strains were kept in our laboratory.
-
Twenty-five 3-week old conventional large White-Duroc crossbred weaned specific-pathogen free piglets were randomly divided into five groups (n = 5). These piglets were confirmed PRRSV negative by ELISA and RT-PCR. Pigs in the first two groups were immunized intramuscularly on day 0 post-vaccination (DPV) with vaccine (PRRSV-MLV, 1 × 106 TCID50/pig). After 4 weeks, pigs in groups 1 and 3 were challenged with homologous PRRSV VR-2332 (1 × 106 TCID50/pig) and pigs in groups 2 and 4 were challenged with heterologous PRRSV JXA1 (25th passage) (1 × 106 TCID50/pig) intramuscularly. Pigs in group 5 served as negative controls and received MEM without virus challenge. Pigs were humanely euthanized at day 14 post-challenge (DPC). All animal experiments were approved by the Institutional Animal Care and Use Committee at National Research Center for Veterinary Medicine and conventional animal welfare regulations and standards were adhered to.
-
Blood was collected on 28 DPV and 14 DPC. Fresh blood samples were directly subjected to flow cytometry analysis. Peripheral blood mononuclear cells (PBMCs) were isolated from heparinized blood samples by Ficoll-Hypaque gradient centrifugation using Histopaque®-1077 (SigmaAldrich, St. Louis, MO). PBMCs were used for ELISpot assay.
-
In brief, half a million PBMCs were plated in enriched RPMI (Thermo Fisher Scientific, MA) in a 96-well multiscreen plate (Millipore, Billerica, MA) which had been precoated overnight with capture IFNγ mAB (BD pharMingen, San Diego, CA). PBMCs were re-stimulated with 1 × 105 TCID50 inactivated PRRSV VR2332 or JXA1 for 24 h at 37 ℃. IFNγ-secreting cells were detected using a biotinylated anti-pig IFNγ detection antibody (clone P2C11) and visualized using an immunospot image analyzer (Cellular Technology, Cleveland, OH). Data are presented as mean numbers of antigen-specific IFNγ-secreting cells per 106 PBMCs from duplicate wells of each sample.
Four-color or three-color flow cytometric assays were developed to identify different lymphocyte subsets. In the four-color cell staining protocol, FITC-labeled mouse antipig CD3ε (clone BB23-8E6-8C8), PerCP-CyTM5.5-labeled mouse anti-pig IFNγ (clone P2G10), PE-labeled mouse anti-pig CD4α (clone 74-12-4), and APC-labeled mouse anti-pig CD8α (clone MIL12) antibodies were used. In brief, 1 μL Brefeldin A (Biolegend, San Diego, CA), which blocks protein secretion and leads to IFNγ accumulation in the Golgi complex, was added to cells. Cells were permeabilized with permeabilization/fixation buffer (Biolegend, San Diego, CA) the stained with PerCP-CyTM5.5- labeled mouse anti-pig IFNγ. After rinsing in phosphate buffered saline three times, cells were stained with other three antibodies. Different lymphocyte populations identified based on cell surface marker phenotype were T-helper cells (CD3+CD4+CD8-), cytotoxic T lymphocytes (CD3+CD4-CD8+), T memory/Th cells (CD3+CD4+ CD8+), and NK cells (CD3-CD4-CD8+). The three-color cell staining protocol was used to identify the γδ T cell population. PerCP-CyTM5.5 mouse anti-pig IFN-c (clone P2G10), FITC-labeled mouse anti-pig CD8α (clone 76-2- 11), and mouse anti-pig γδ T cell (TcR1N4, purchase from VMRD. Pullman, WA) antibodies were used. The staining protocol used was for four-color flow cytometric assays as described. Unless otherwise noted, all commercial antibodies listed were purchased from BD Biosciences. Cells were acquired using a FACS Calibur (BD Biosciences) flow cytometer. Frequencies of individual lymphocytes were analyzed by one million events using FlowJo software (Tree Star, Inc., OR, USA).
-
Total RNA was extracted from serum collected on 7 DPC and 14 DPC. One-step SyBR Green real-time PCR (BioRad, Hercules, CA) was performed to evaluate the PRRSV ORF7 expression level. For quantification, VR2332 PRRVS RNA of a known TCID50 was serially diluted and used to generate a standard curve. The virus quantities in unknown samples were determined by linear extrapolation of the Ct value plotted against the standard curve (Li et al. 2013).
-
All data are expressed as the mean value from five pigs ± SEM. The percentage difference in IFNγ-secreting cells between two PRRSV challenge strains was determined by student t test (Prism5.0, GraphPad Software, SanDiego, CA). Differences were considered statistically significant when P < 0.05.
Cells and Viruses
Pigs, Vaccination, and Viral Challenge Study
Collection of Blood Samples for Analysis
ELISpot Assay and Flow Cytometry Analysis
Analysis of Viremia Using Real-Time PCR
Statistical Analysis
-
Brefeldin A blocks protein secretion and leads to the accumulation of IFNγ in the Golgi complex (Li et al. 2013). Whole blood samples, from naïve or vaccinated pigs, were stimulated with inactivated PRRSV to measure IFNγ-production in the presence of Brefeldin A. Blood samples without any stimulation served as a control. The four-color flow cytometric assay was developed for the phenotypic characterization of porcine IFNγ-producing lymphocytes. No fluorescence signal was detected in unstimulated cells (Supplementary Figure S1A ). After PRRSV stimulation, naïve porcine blood had a low frequency (0.09% ± 0.03%) of IFNγ-producing cells in CD3- gated lymphocytes (Supplementary Figure S1B). The frequency of IFNγ-producing cells in blood from PRRSVvaccinated pigs (0.42% ± 0.12%) was higher than that observed in naïve porcine blood (Supplementary Figure S1C, left two figures). CD3+ IFNγ-producing cells were further gated by CD4 and CD8 to delineate the percentage of IFNγ-producing in T-helper (CD3+CD4+), cytotoxic T (CD3+CD8+), and Th/memory (CD3+CD4+ CD8+) cell populations (Supplementary Figure S1C right top). CD3- IFNγ-producing cells were further gated by CD4 and CD8 to delineate the percentage of IFNγ-producing in the NK (CD3-CD4-CD8+) cell population (Supplementary Figure S1C right bottom). γδ T cells are another important porcine IFNγ producing T lymphocyte resource. To determine the proportion of γδ T cells contributing to IFNγ-producing after PRRSV stimulation, CD8+ and γδ+ cell populations were gated and defined as γδ T cells in IFNγ-producing lymphocytes (Supplementary Figure S1D).
-
Whole blood samples were collected at 28 DPV and PBMCs were isolated to perform an ELIspot assay to quantify IFNγ-secreting cells. Only PRRSV-MLVvaccinated pigs developed PRRSV-specific IFNγ-secreting cells at 28 DPV (Fig. 1). As expected, the frequency of PRRSV-specific IFNγ-secreting cells in naïve pigs was negligible. There was no significant difference in the frequency of IFNγ-secreting cells following PBMC stimulation with PRRSV VR2332 or JXA1 strains.

Figure 1. Frequency of IFNγ-secreting cells after vaccination determined by ELIspot assay. Peripheral blood mononuclear cells (PBMCs) from Naïve or vaccinated pigs were collected at 28 DPV and re-stimulated with inactivated VR2332 or JXA1 PRRSV to measure the frequency of IFNγ-secreting cells. Each bar represents the average number of IFNγ-secreting cells per million PBMCs of five pigs ± SEM. NS indicates no statistical difference
-
Different PBMC T cell subsets were analyzed at 28 DPV. The percentages of all T cell subsets were increased in vaccinated pigs compared with unvaccinated pigs (Table 1). Since T-helper cells, cytotoxic T cells, Th/memory cells, NK cells, and γδ T cells were reported to be responsible for IFNγ generation, we next explored the contribution of the different cell subsets to IFNγ-producing after PRRSV-MLV vaccination. Of the different lymphocyte subsets, T-helper cells accounted for over 30% of total IFNγ-secreting cells (Fig. 2). γδ T cells are T cell subsets unique to porcine blood and are important resources for IFNγ-producing, especially in young pigs. γδ T cells accounted for approximately 13% of IFNγ-producing following PRRSV vaccination (Fig. 2). CTLs and Th/memory cells accounted for 6% and 7% of total IFNγsecreting cells, respectively. NK cells accounted for 3% of total IFNγ-secreting cells.

Table 1. Frequency of T cell subsets in pigs after vaccination. Peripheral blood mononuclear cells (PBMCs) were isolated from blood collected from pigs at 28 DPV

Figure 2. Percentage of different lymphocyte subsets contributing to IFNγ-producing after PRRSV-MLV vaccination (1 million cells were used). Whole blood samples from PRRSV-MLV vaccinated pigs were collected at 28 DPV for flow cytometric assay. Each bar represents the average percentage of IFNγ-secreting cells of five pigs ± SEM
-
All pigs were challenged with homologous VR2332 (shares 98.9% genome similarity with PRRSV-MLV) or heterologous JXA1 (shares 89.4% genome similarity with PRRSVMLV) at 28 DPV. Different PBMC T cell subsets were also analyzed at 14DPC.Exceptfor T-helper cells(in theVR2332- challenge scenario) and γδ T cells, the percentages of all T cell subsets were decreased in unvaccinated pigs irrespective of whether challenged with VR2332 or JXA1 (Table 2). In contrast, the percentages of all T cell subsets were increased in vaccinated pigs. PBMCs from PRRSV-challenged pigs were stimulated with VR2332 or JXA1 PRRSVs before performing ELIspot assay. At 14 DPC, the IFNγ-secreting cells population increased significantly in PRRSV-MLV vaccinated pigs (MLV + VR2332 and MLV + JXA1 groups) compared with that in unvaccinated pigs (Naïve +VR2332 and Naïve +JXA1 groups) (Fig. 3). There was a higher frequency of IFNγ-secreting cells in VR2332-challenged pigs following PBMCs re-stimulation with homologous VR2332 virus than with heterologous JXA1 virus (within MLV + VR2332 group). Compared to pigs challenged with VR2332, JXA1- challenged pigs developed more IFNγ-secreting cells (MLV + VR2332 group vs. MLV + JXA1 group). However, there was no significant difference in IFNγ-producing following PBMC re-stimulation with VR2332 or JXA1 in this pig group (MLV + JXA1 group).

Table 2. Frequency of T cell subsets in pigs after viral challenge. Peripheral blood mononuclear cells (PBMCs) were isolated from blood collected from pigs at 14 DPC

Figure 3. Frequency of IFNγ-secreting cells analyzed by ELIspot assay after viral challenge. Naïve or PRRSV-MLV vaccinated pigs were challenged with VR2332 or JXA1 PRRSV. PBMCs from PRRSVchallenged pigs were collected at 14 DPC, and re-stimulated with inactivated VR2332 or JXA1 PRRSV before ELIspot assay. Each bar represents the average number of IFNγ-secreting cells per million PBMCs of five pigs ± SEM. Asterisk denotes a statistically significant difference (P < 0.05). NS indicates no statistical difference
-
To determine how the different cell populations contributing to IFNγ-producing when vaccinated pigs re-encountered PRRSVs, the percentages of IFNγ-secreting cells of different populations were analyzed. T-helper cells still accounted for the largest cell proportion after viral challenge, followed by γδ T cells and Th/memory cells (Fig. 4). The percentages of IFNγ-secreting cells within each cell population were compared following VR2332 or JXA1 challenge. The percentage of Th/memory cells for total IFNγ-secreting cells was significantly higher in VR2332-challenged pigs than in JXA1-challenged pigs. There was no significant difference as for the percentage of other cell populations for total IFNγ-secreting cells after VR2332 or JXA1 virus challenges.
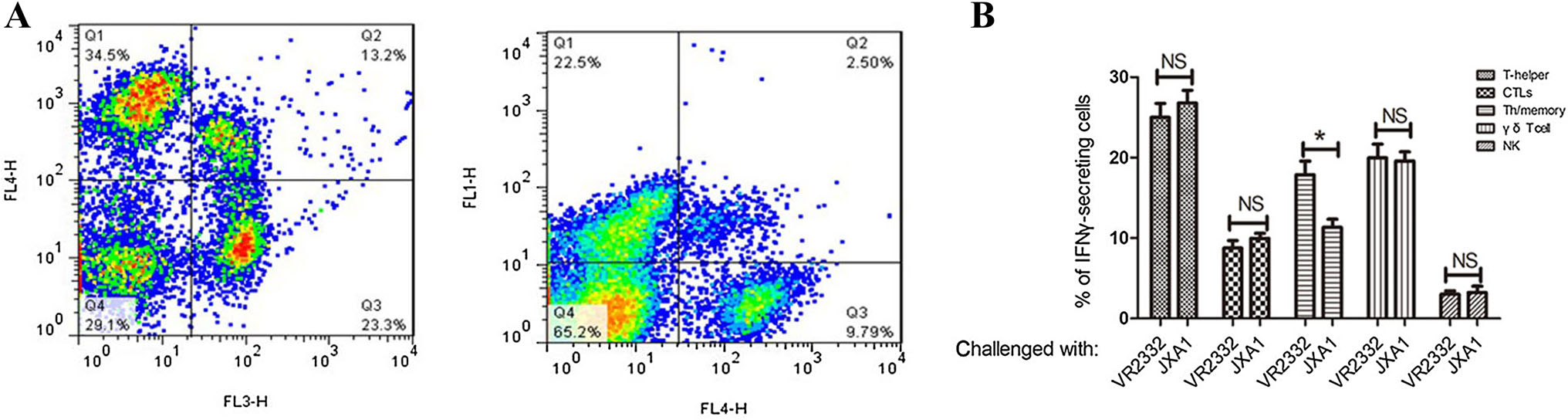
Figure 4. Percentage of different cell populations that contributed to IFNγ-producing after virus challenge as analyzed by flow cytometry. PRRSV-MLV vaccinated pigs were challenged with VR2332 or JXA1 PRRSV. Whole blood samples were collected for flow cytometric analysis at 14 DPC. (A) Representative flow cytometry profile of lymphocytes after viral challenge. CD3+ gated lymphocytes expressing CD4+ (FL3) and CD8+ (FL4) are shown (left). Lymphocytes expressing CD8+ (FL1) and γδ T+ (FL4) are shown (right). (B) Contribution of different cell populations to IFNγ-producing. Each bar represents the average percentage of IFNγ-secreting cells of five pigs ± SEM. Asterisk denotes a statistically significant difference (P < 0.05). NS indicates no statistical difference
-
To explore the correlation between IFNγ-producing and virus load, serum sample virus titer was measured using real-time PCR. MLV-vaccinated pigs had decreased serum sample virus titers 7 DPC (Fig. 5). On DPC 14, the virus titer in vaccinated pigs was significantly lower than that of unvaccinated pigs, which illustrated the efficacy of vaccination. Compared to the pigs in the heterologous challenge group (MLV + JXA1), the viremia of pigs in the homologous challenge group (MLV + VR2332) was undetectable. No clinical symptoms were exhibited by any vaccinated pigs. By contrast, unvaccinated pigs challenged with VR2332 had a transient fever (2–5 DPC) and no other clinical symptoms were observed. Unvaccinated pigs challenged with JXA1 had lasting fever from 2 to 10 DPC and showed transient clinical symptoms including dyspnea, coughing, and shivering. All pigs that survived to the end of study were terminated at 14 DPC.
Flow Cytometry Setup to Differentiate Subsets of Lymphocytes
Frequency of IFNγ-Secreting Cells After PRRSVMLV Vaccination
Contribution of Different Lymphocyte Subsets to IFNγ Production After PRRSV-MLV Vaccination
Frequency of IFNγ-Secreting Cells After Homologous or Heterologous PRRSV Challenge
Contribution of Different Lymphocyte Subsets to IFNγ Production After PRRSV Challenge
Viremia Profiles at 7 DPC and 14 DPC
-
IFNγ plays pivotal roles in host cell-mediated immunity including in facilitating complete viral clearance and providing protection when reencountering the same pathogen in PRRSV infection (Toyoda et al. 2012). In human and rodents, IFNγ is produced by T lymphocytes and NK cells (Keane et al. 2012). Porcine peripheral T lymphocytes have two unique subsets: Th/memory and γδ T cells. To date, Th, CTLS, Th/memory, and γδ T lymphocytes have been reported to produce IFNγ after virus infection. Previous studies indicated that PRRSV-specific cell-mediated immunity, assessed by IFNγ-secreting cells, contributes to complete virus clearance (Costers et al. 2009). However, the nature of porcine IFNγ-producing lymphocytes following PRRSV infection has not yet been elucidated. In this study, we first explored the contribution of different lymphocyte subsets to IFNγ-producing after PRRSV vaccination and compared the phenotypes of IFNγ-secreting cells following homologous and heterologous virus challenge.
The multiple-color flow cytometric assay was developed to delineate the percentage of porcine peripheral blood lymphocytes that produce IFNγ upon stimulation with PRRSV. ELIspot and flow cytometric assay results showed that unvaccinated pigs did not develop PRRSV-specific IFNγ-secreting cells. In vaccinated pigs, the major proportion of IFNγ-producing cells after vaccination were Th cells, followed by γδ T cells, Th/memory cells, and CTLs (Fig. 2). After viral challenge, increased IFNγ-producing cell proportions were observed in Th/memory and γδ T cell populations, although Th cells remained the highest proportion of IFNγ-secreting cells (Fig. 4). These results are consistent with those of a previous report which showed that Th cells constitute a higher proportion of IFNγ-secreting cells when vaccinated pigs are challenged with VR2332 and a local strain of KS-06 (Li et al. 2014). Additionally, the similar frequencies of different T lymphocytes in response to VR2332 and JXA1 PRRSV challenge suggest that different PRRSV challenge does not affect T cell populations.
CD4+CD8+ double positive T cells, exhibiting features of memory cells, play a particularly important role in IFNγ production when pigs reencounter PRRSV. The capacity of CD4+CD8+ T cells to produce high levels of IFNγ has been reported previously (De Bruin et al. 2000). The increased proportion of Th/memory cells contributing to IFNγ-producing was assumed to result from increased ability to secret IFNγ because the percentage of Th/memory cells in lymphocytes before and after virus challenge remained stable. Compared to JXA1 heterologous challenge pigs, Th/memory cells generated in VR2332-challenged pigs were responsible for a higher proportion of IFNγ-producing (Fig. 4). PRRSV structural Gp5 and M proteins were reported to induce IFNγ-producing after PPRSV infection (Jiang et al. 2006). The amino acid of these two structural proteins in MLV and VR2332 share more than 99.2% similarity, while JXA1 shares only 89.4% similarity with MLV. This could explain the disparity of IFNγ production in Th/memory cells when vaccinated pigs were challenged with different PRRSV strains.
The proportion of γδ T cells that contributed to IFNγ production increased after virus challenges. γδ T lymphocytes are not linked to major histocompatibility complex (MHC) or non-MHC genes, and do not have memory characteristics (Riganti et al. 2012). Therefore, the increased contribution to IFNγ-producing by γδ T cells may simply be due to increased numbers of these cells in blood. Also, a similar percentage of γδ T cells contributed to IFNγ-producing after VR2332 or JXA1 challenge, illustrating that there was no phenotypic memory response (Fig. 4).
After JXA1 PRRSV re-stimulation, pigs in the MLV + JXA1 group had higher proportions of IFNγ-secreting cells in their PBMCs (Fig. 3). However, the viremia level in the pigs of this group was significantly higher than in the pigs in the MLV + VR2332 group. In Xiao's study, they reported that the level of IFNγ expression after PRRSV infection was variable and did not correlate with virus load (Xiao et al. 2004). Moreover, other immune factors, such as IFNa, have recently been shown to alter adaptive immune responses to PRRSV (Zhang et al. 2012).
In summary, we used the ELIspot assay and flow cytometry to analyze the percentages of different lymphocyte subsets contributing to IFNγ-producing after PRRSV vaccination and challenge. We then delineated the change profiles of different T lymphocyte subset following homologous and heterologous PRRSV challenges. Our results will extend our understanding of the roles of IFNγ in protective immunity against PRRSV infection. Moreover, our results will provide useful information to elicit superior IFNγ-mediated CMI response and better crossprotective immunity in future PRRSV vaccine design.
-
This work was supported by Grant from The National Natural Science Foundation of China (NSFC, Grant No. 31490601), National Key Research and Development Program (Grant No. 2016YFD0500703), Major Science and Technology Projects in Henan Province (Grant No. 171100110200), and Luoyang Heluo Talent Plan (Dr. Kegong Tian).
-
JS and KT design the study. XL, ZP, YB and LW performed the experiments. XL analyzed the data and drafted the manuscript. XL, JS and KT wrote the manuscript. All authors read and approved the final manuscript.
Acknowledgements
Author Contributions
-
The authors declare that they have no conflict of interest.
-
The whole study was approved by the Animal Ethics Committee of National Research Center for Veterinary Medicine in China (Permit No. NVC-2017-018). All institutional and national guidelines for the care and use of laboratory animals were followed.
Conflict of interest
Animal and Human Rights Statement
-

Figure 1. Representative flowcytometry profiles of different IFNγ-secreting cell populations. (A) Naïve pig control without porcine reproductive and respiratory syndrome virus (PRRSV) re-stimulation. (B) Naïve pig blood with PRRSV re-stimulation. (C) The IFNγ-secreting CD4+, CD8+, CD4+CD8+ and NK cell populations (four-color straining protocol). PRRSV-vaccinated pig blood was re-stimulated with inactivated PRRSV. Lymphocytes were first gated by CD3 (FL1) and IFNγ (FL3) to delineate the CD3+ IFNγ-secreting population and CD3- IFNγ-secreting population. The above two populations were further gated by CD4 (FL2) and CD8 (FL4) to delineate IFNγ-secreting CD4+, CD8+, CD4+CD8+, and NK cell populations. (D) IFNγ-secreting γδ T cell population (three-color straining protocol). IFNγ-secreting cells were first gated (FL3). IFNγ+ lymphocytes were further gated by CD8 (FL1) and γδ (FL4)

















 DownLoad:
DownLoad: