HTML
-
Papillomaviruses (PVs) are a family of small double-stranded circular DNA viruses with more than 200 genotypes (Doorbar 2016). Human papillomaviruses (HPVs) have been demonstrated to be responsible for causing several human cancers and genital warts (Moody and Laimins, 2010). The persistent infection of high-risk human papillomavirus (hr-HPV) is closely related to the pathogenesis of cervical cancer and many other cancers. Indeed, HPVs account for more than 30% of all infection-associated cancers in humans (zur Hausen 2009). Preventive vaccines including 2-valent, 4-valent and 9-valent vaccines against HPV-associated cervical cancer are now available, which prevent an estimated 92% of the cancers attributable to HPV types (Frazer and Levin 2011; Huh et al. 2017; Zhang et al. 2020a). Considering that the preventive vaccines are unable to wipe out the existing HPV viruses in already infected people, scientists have been focusing on developing therapeutic HPV vaccines that have reached the clinical trial phase in cervical cancers and diseases. Many therapeutic HPV vaccines tested in clinical trials show the potential use of the vaccines as safe and effective pharmacological tools (Ding et al. 2010; Zhang et al. 2020a; Zhao et al. 1998; Zottnick et al. 2020). Recently, published studies have revealed that HPV minor capsid L2 proteins play a crucial role in the early stage of HPV infection (DiGiuseppe et al. 2015; Gagnon et al. 2015; Zhang et al. 2018, 2020b). The C terminus of HPV L2 proteins contains a short sequence of basic amino acids serving as a cell-penetrating peptide (CPP) (Campos 2017; DiGiuseppe et al. 2015; Zhang et al. 2018). The CPP binds to a cytoplasmic trafficking factor, retromer, thereby sorting the virus into the retrograde pathway for properly trafficking the incoming virus to the nucleus, the site of viral gene expression and DNA replication (Campos 2017; DiGiuseppe et al. 2015; Zhang et al. 2018). Thus, it appears that there is a need for specific drugs for targeting the CPP of L2 proteins to prevent HPV infection potentially leading to the development of HPV therapy against HPV-induced cancer.
PVs have also been identified in most domestic animals such as bovines (BPVs), canines (CPVs), goats (Capra hircus papillomavirus-ChPVs), equines (Eqnus caballus papillomavirus EcPVs) (Modolo et al. 2017). BPVs are the most typical animal PVs, which are a cosmopolitan virus, worldwide distributed, independently of the level of expertise on livestock exploration (Araldi et al. 2017). BPVs containing 15 types have been classified into 3 separate genera: Deltapapillomavirus, Epsilonpapillomavirus, and Xipapillomavirus, which are causative agents of benign and malignant tumors in cattle, such as cutaneous papilloma, fibropapilloma, urinary bladder and esophageal cancers (Borzacchiello and Roperto 2008; Campo 2003; Munday et al. 2015). BPV-1, BPV-2, and BPV-13, for example, are classified in the Deltapapillomavirus genus and induce fibropapilloma (Munday et al. 2015; Thomson et al. 2015). Since 1970, BPV-1 has been used as a prototype papillomavirus to study PV biology and oncology (Araldi et al. 2017; Borzacchiello and Roperto 2008; Borzacchiello et al. 2006; Koller and Olson 1972). As a model system, BPV-1 has been used to study the regulation of DNA replication in higher eukaryotes (Schvartzman et al. 1990). In addition, transcription of BPV-1 viral RNAs has been previously reported in BPV-1 transformed cells and in warts (Amtmann and Sauer, 1982). The study of using BPV has greatly improved our understanding of HPV-associated carcinogenesis (Munday et al. 2015). We and others have previously developed an in vitro Saccharomyces cerevisiae (budding yeast) system, which is permissive for viral DNA replication (e.g. BPV-1 and different types of HPVs) (Angeletti et al. 2002; Zhao and Frazer 2002a, b). We discovered that S. cerevisiae was infected with BPV-1 virions isolated from the bovine papilloma leading to not only the replication of BPV-1 genomic DNA, but also the production of infectious virus-like particles (Zhao and Frazer 2002a, b). The use of S. cerevisiae for the replication of both plant and animal viruses was a breakthrough (Navarro et al. 2004).
Several studies have reported the replication patterns of PV genome in different systems by means of different techniques such as Southern blot, chromogenic in situ hybridization (CISH) and PCR using specific and/or degenerate primers (Araldi et al. 2015; Melo CA and Melo SA, 2014; Melo et al. 2015). In addition, using the restriction fragment length polymorphism of PCR products (PCR-RFLP) allows to identify BPV type (Carvalho et al. 2013), since this method shows a correlation of 95% with the results obtained using DNA sequencing (Carvalho et al. 2013; Kawauchi et al. 2015). However, the above methods cannot be used to characterize the replication mode and intermediates of the viral DNA that persists as extrachromosomal plasmids in eukaryotic cells and neither to resolve the initiation, elongation, and termination of DNA replication associated with distinct, nonlinear DNA structures. Bell and Byers first developed two-dimensional agarose gel electrophoresis (2-DAGE) to study the shape of DNA recombination intermediates (Bell and Byers 1983). The 2-DAGE includes a first and a second (run perpendicular to the first) dimension agarose gel electrophoresis. The first dimension is a conventional separation of DNA by molecular size. The second dimension is to separate the molecules mainly on the basis of the DNA shape. Since then, 2-DAGE has been proved to be highly advanced and useful for studying the complex topological problem of DNA replication. The rationale and advances of 2-DAGE for DNA replication study include: (1) mapping origins and termination sites of DNA replication; (2) investigating the efficiency of different origins and the progress of DNA replication forks along DNA fragments; (3) determining the activity level of putative replication origin-containing sequences; (4) analyzing replication timing, fork progression, fork pausing, fork stalling and collapse, termination, and recombinational repair; and (5) resolving the nonlinear, replicating DNA molecules from the linear, nonreplicating molecules (Dandjinou et al. 2006; Lemacon et al. 2017; Makovets 2013; Olavarrieta et al. 2002; Quinet et al. 2017). Indeed, Schvartzman et al. identified extrachromosomal forms of BPV-1 DNA replication in viral DNA-transformed ID13 cells by 2-DAGE (Schvartzman et al. 1990). We have also observed a single replication bubble of BPV-1 DNA in short-term virion-infected S. cerevisiae cultures by 2-DAGE (Zhao and Frazer 2002a).
Although multiple HPV genomes replicate stably in S. cerevisiae for a long-term (up to 75 h), but they differ in replication efficiency (Rogers et al. 2008). The patterns of PV DNA replication and the replication intermediates in S. cerevisiae remain unknown. Thus, we proposed to study the episomal replication patterns and intermediates of BPV-1 DNA in long-term virion-infected S. cerevisiae cultures (up to 108 days) by means of one- and two-dimensional gel electrophoresis and Southern blot hybridization. As an attempt to use BPV-1 model, we studied the regulation of DNA replication in a latently virion-infected single-celled eukaryotic organism. The obtained data have highlighted the characteristics of BPV-1 genomic DNA giving some insights of virus infection in S. cerevisiae system although the exact mechanism of BPV-1 DNA replication remains unclear.
-
BPV-1 virions used for S. cerevisiae protoplast infection were prepared from bovine papilloma as reported previously (Zhao and Frazer 2002a, b). Virions in suspension were dialyzed against 0.15 mol/L phosphate-buffered saline (PBS) (pH 7.4) for 30 min and then used to infect S. cerevisiae protoplasts.
-
BPV-1-infected S. cerevisiae cells were grown in vitro for a long-term culture, similar to our previous description (Zhao and Frazer 2002a, b ), with some modifications. Briefly, S. cerevisiae protoplasts (10 mL; 5 × 107 cells/mL) infected with 0.6 µg of BPV-1 virions was grown in S. cerevisiae medium containing 0.8 mol/L sorbitol and 0.2 mol/L glucose on a shaker with gentle agitation at 28 ℃ in the dark for three separate long time courses (82, 85 and 108 days). S. cerevisiae protoplasts without infection of BPV-1 virions were cultured under the same condition as a negative control. Ten to twelve time points were designed to collect S. cerevisiae cells for analysis of viral DNA replication. At each time point, 5 mL of S. cerevisiae cells was collected and allocated for Hirt supernatant DNA (Hirt DNA), RNA and protein preparation, with 5 mL of fresh medium without sorbitol added for continuous culture.
-
BPV-1-infected S. cerevisiae cultures were used for the preparation of Hirt DNA as described. Digestion of S. cerevisiae cells with enzyme was the same as that for S. cerevisiae protoplast preparation. The digested S. cerevisiae cells were washed with 1 mol/L sorbitol and lysed in 400 µL of lysate buffer (10 mmol/L Tris–HCl, pH 7.5, 10 mmol/L EDTA, and 0.2% Triton X-100) at 25 ℃ for 10 min. Then 100 µL of 5 mol/L NaCl was added to the lysate, and the mixture was frozen at −20 ℃ for 40 min. The frozen lysates were thawed at 25 ℃ for 20 min. The resultant supernatants containing viral DNA (Hirt DNA) were incubated with 100 µg of proteinase K at 37 ℃ for 1 h. The Hirt DNA was extracted with Tris-buffered phenol twice and chloroform once and then precipitated by ethanol. The Hirt DNA was then used for agarose gel electrophoresis and Southern blot hybridization.
-
Hirt DNA (10 µg) partially digested with Hind Ⅲ as previously reported (Zhao and Frazer 2002a, b ) was electrophoresed on a 1% one-dimensional agarose gel electrophoresis (1-DAGE), then blotted onto nylon membranes, and hybridized with a 32P-labeled BPV-1 DNA probe (Zhao and Frazer 2002a).
-
Hirt DNA (15 µg) without enzyme digestion as reported by Schvartzman and colleagues (Schvartzman et al. 1990) was separated on a neutral/alkaline 2-DAGE. The Hirt DNA was electrophoresed on a first-dimension agarose gel (0.4%) in TAE buffer (40 mmol/L Tris-acetate, 2 mmol/L EDTA, pH 8.0) at 1.5 V/cm for 20 h. The DNA lanes were run on a second-dimension agarose gel (1.0% agarose in sterile distilled H2O) in alkaline electrophoresis buffer (40 mmol/L NaOH, 2 mmol/L EDTA) at 1.5 V/cm for 24 h and then blotted onto nylon membranes. The DNA blots were hybridized with a 32P-labeled BPV-1 DNA probe that was a linearized BPV genome to detect all the replication intermediates (Zhao and Frazer 2002a, b ). According to previous studies (Flores and Lambert 1997; Sakakibara et al. 2013; Schvartzman et al. 1990), the replication mode and intermediates of the BPV-1 DNA in S. cerevisiae were analyzed and identified. The replication intermediates included single-stranded DNA (1ss), double-stranded DNA (2ss), heterogeneous double-stranded DNA (hds), linear forms (lin) and a series of oligomers. Replication intermediates also included recombinant-dependent replication (RDR), rolling circle replication (RCR) and conspicuous multimeric circular ssDNA (cms). A diagram drawn from the visualized Southern blot hybridization results was used to explain in details the identified replication intermediates of BPV-1 DNA occurred in S. cerevisiae cells in Result section.
BPV-1 Virion Preparation
Cultures of BPV-1-Infected S. cerevisiae Cells and Sample Collection
Hirt DNA Preparation
One-Dimensional Agarose Gel Electrophoresis and Southern Blot Hybridization
Two-Dimensional Agarose Gel Electrophoresis and Southern Blot Hybridization
-
We previously demonstrated that BPV-1 DNA could be detected in a small volume (2 mL) of S. cerevisiae cells infected with BPV-1virions at day 55 and 75 post-infection by 1-DAGE and Southern blot hybridization of Hirt DNA (Zhao and Frazer 2002a, b ). Thus, to confirm further that BPV-1 DNA could be persistently present in the BPV-1-infected S. cerevisiae cells in a relatively large volume (10 mL) cultured for a longer period at 28 ℃, we carried out three separate long term BPV-1 virion-infected S. cerevisiae culture experiments up to 108 days. It was observed that the BPV-1-infected S. cerevisiae cells grew slowly from day 23/34 to day 45 and most of the cells showed unhealthy morphology (Supplementary Fig. S1). We prepared Hirt DNA from the long term BPV-1-infected S. cerevisiae cultures at various time points to investigate whether and how BPV-1 DNA was present by Southern blot analysis. The Hirt DNA was subjected to partial digestion with HindIII, which should cut BPV-1 DNA at a single site. The partial-digested Hirt DNA hybridized with BPV-1 DNA revealed that BPV-1 DNA was detected in all three long-term BPV-1-infected S. cerevisiae cultures (Fig. 1). The present forms of BPV-1 DNA in the long-term BPV-1-infected S. cerevisiae cultures varied distinctly from the early stage to the late stage (Fig. 1). At the early stage (day 3 to day 16/23), three bands of the BPV-1 DNA were observed to correspond to nicked circular, linear, and supercoiled monomeric BPV-1 DNA although the signals of nicked circular form were very weak. At the middle and late stages (day 23 to day 82), nicked circular form was undetectable, while signals of the other two forms decreased significantly at day 23/34 then at a constant level. Compared with the nicked circular and supercoiled monomeric forms, the linear BPV-1 DNA had the strongest signals over the time period (Fig. 1A), which could be attributed to the partial digestion of HindIII. The present patterns of BPV-1 DNA detected in the three long-term BPV-1-infected S. cerevisiae cell cultures were generally similar, but the signals of nicked circular and supercoiled forms differed (Fig. 1). The results confirmed that S. cerevisiae infected with BPV-1 virions could maintain episomal BPV-1 DNA for a long-term period. The results also suggested that the viral genome was actively amplified in the BPV-1-infected S. cerevisiae cells at the early stage, and then replicated at a low and constant copy number at the middle and late stages to establish persistent infection in the S. cerevisiae cells.

Figure 1. BPV-1 DNA detected by Southern blot hybridization in BPV-1 virion-infected S. cerevisiae culture in three-time course experiments: day 0 to day 82 (A), day 0 to day 85 (B), day 0 to day 108 (C). S. cerevisiae cells (10 mL; 5 × 107cells/mL) infected with 0.6 µg of BPV-1 was cultured at 28 ℃ in the dark. At each time point, 5 mL of S. cerevisiae cells was collected for Hirt DNA preparation, with 5 mL of fresh medium added for continuous culture. 10 µg of Hirt DNA without partial digestion of HindIII was electrophoresed on a 1% agarose gel and blotted onto a nylon membrane. Blots were hybridized with BPV-1 DNA using [α-32P] dCTP at 3000 Ci/mmol and exposed using Kodak BioMax film at −70 ℃ for 24 h. N, nicked circular BPV-1 DNA; L, linear BPV-1 DNA; S, supercoiled monomeric BPV-1 DNA
-
We have previously confirmed that BPV-1 DNA detected in the short term BPV-1-infected S. cerevisiae culture at one-time point was replicating, with a single replication bubble observed, which was a typical replication pattern by 2-DAGE and Southern blot hybridization (Zhao and Frazer 2002a). Here, the Hirt DNA samples without HindIII digestion were used to carry out 2-DAGE and Southern blot hybridization to analyze newly synthesized viral genomic DNA. Most of the newly replicated duplex DNA was converted into small molecules: the replication intermediates, which served as the templates for additional replication or transcription. The generation of the replication intermediates of viral DNA over the time course could be divided into three culture stages: early (day 3–16), middle (day 23–34/45), and late-stage (day 45–82). At the early culture stage (day 3, 7, 11 and 16), much more multiple replication intermediates such as the linear monomers, dimers, trimers, and higher oligomers were generated and shown as covalently closed circles (CCC1, CCC2, CCC3, CCC4 and CCC5) and open circles (OC1, OC2, OC3, OC4) (Fig. 2). The cccDNAs are the most important replication intermediates that serve as the main nuclear transcription template for producing all viral RNAs in the viral life cycle. Furthermore, the replication intermediates including 1ss, 2ss and lin consistently occurred at this stage (Fig. 2). Also, other replication intermediates such as hds, cms, RCR and RDR intermediates were detected (Fig. 2). Although so many different replication intermediates were detected, the occurrences of these intermediates except for 1ss, 2ss and lin forms were significantly different from one-time point to another time point. Generally, significantly more replication intermediates were detected in the early culture stage (day 3, 7, 11 and 16) than those in the middle culture stage (day 23 and 34) (Supplementary Fig. S2) and late culture stage (day 45, 55, 72 and 82) (Fig. 3). In fact, only 1ss, 2ss, lin and hds forms were present, other replication intermediates were scarcely detected in the middle culture stage (day 23 and 34) (Supplementary Fig. S2). It appeared that much less replication intermediates occurred in the middle stage were associated with poor growth of the BPV-1-infected S. cerevisiae cells (Supplementary Fig. S1) and poor replication status of the viral DNA (Fig. 1). In the late culture stage, replication intermediates such as CCC1, CCC5, OC1, OC2, OC3, OC4 and cms could be detected, but not as many as those observed at the early culture stage (Figs. 2 and 3). In addition, Y-shaped replication intermediates were observed at several time points in both early and late stages (Figs. 2 and 3).

Figure 2. Analysis of viral DNA replication intermediates in BPV-1 virion-infected S. cerevisiae cells culture for 3, 7, 11 and 16 days by means of two-dimensional gel electrophoresis. 15 µg Hirt DNA was electrophoresed on a first-dimension agarose gel (0.4%) in TAE buffer (40 mmol/L Tris–acetate, 2 mmol/L EDTA, pH 8.0) at 1.5 V/cm for 20 h. The DNA lanes were run on a second-dimension agarose gel (1.0% agarose in sterile distilled H2O) in alkaline electrophoresis buffer (40 mmol/L NaOH, 2 mmol/L EDTA) at 1.5 V/cm for 24 h and then blotted onto nylon membranes. The DNA blots were hybridized with a 32P-labeled BPV-1 DNA probe. Replication intermediates include single-stranded DNA (1ss), double-stranded DNA (2ss), heterogeneous double-stranded DNA (hds), linear forms (lin) and a series of oligomers. The oligomers contain monomers (1), dimers (2), trimers (3), and higher oligomers (4 or 5), which showed as covalently closed circles (CCC) and open circles (OC). Replication intermediates also include conspicuous multimeric circular ssDNA (cms), rolling circle replication (RCR) and recombinant-dependent replication (RDR) intermediates.
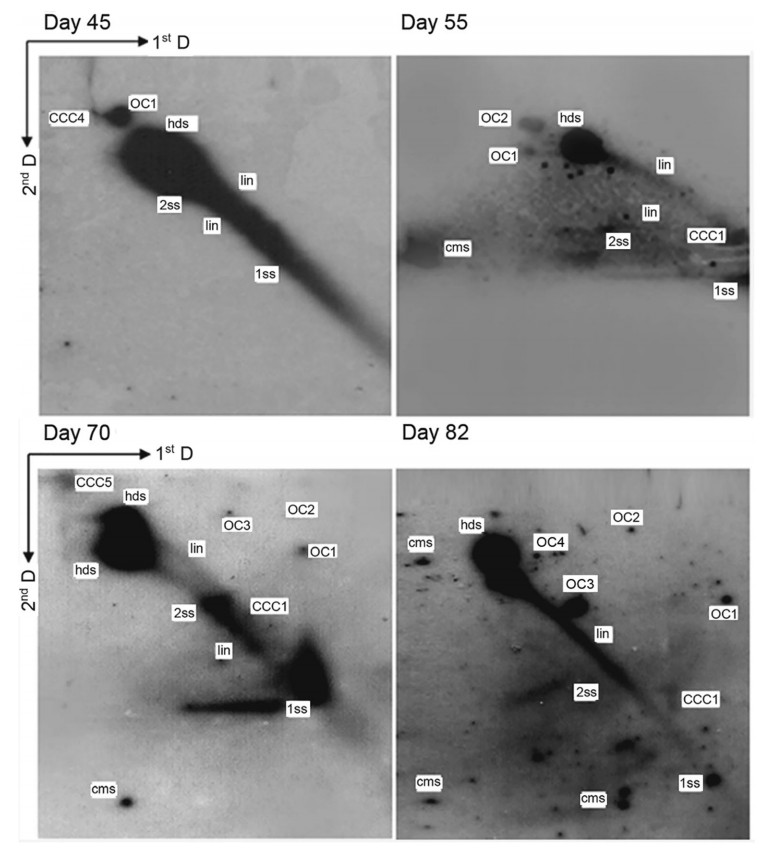
Figure 3. Analysis of viral DNA replication intermediates in BPV-1 virion-infected S. cerevisiae cells culture for 45, 55, 70 and 82 days by means of two-dimensional gel electrophoresis and Southern blot-hybridization analysis. Replication intermediates were detected including not only single-stranded DNA (1ss), double-stranded DNA (2ss), heterogeneous double-stranded DNA (hds), linear forms (lin) but also a series of oligomers such as covalently closed circles (CCC), open circles (OC), conspicuous multimeric circular.
-
We have drawn a diagram to explain the identified replication intermediates of BPV-1 DNA in S. cerevisiae cells mainly based on the day-7 visualized 2-DAGE/Southern blotting result (Fig. 4). Firstly, the identified replication intermediates included 1ss, 2ss, hds, lin and a series of oligomers. The size of 1ss was about 8 kb while the 2ss was approximately 16 kb and hds was over 23 kb. The identified oligomers contained monomers, dimmers, trimers, and higher oligomers, which showed as CCC and OC. Both CCC and OC were detected above the arc of linear molecules, with their occurrences in different sizes associated with the arc numbers of linear molecules. Generally, the OC has shown a slightly higher molecular size than CCC within an arc of linear molecules. Then, the identified replication intermediates included RDR, RCR and cms. RDR is one of HPV DNA replication modes (Sakakibara et al. 2013). RDR occurred in a high molecular size is recognized as important for replication restart and stability, which plays an essential role in the replication cycle of HPV DNA. RCR is a single nicking event on one parental DNA strand, which gives rise to unidirectional replication resulting from the convergence of two replication forks without forming intermediates and termination structures (Flores and Lambert 1997). cms was detected under the arc of linear molecules and generated from the oligomerization of circular single-stranded DNA. cms is independent on the topology of the input DNA and has various molecular sizes due to rapid binding of E1 and slow dissociation (White et al. 2001).

Figure 4. Diagram of replication intermediates of BPV-1 DNA in BPV-1 virion-infected S. cerevisiae cells culture mainly based on the Day-7 visualized 2-DAGE/Southern blotting result (Fig. 2). The replication intermediates include single-stranded DNA (1ss), double-stranded DNA (2ss), heterogeneous double-stranded DNA (hds), linear forms (lin). The replication intermediates also include multiple oligomers that are covalently closed circles (CCC), open circles (OC), recombinant-dependent replication (RDR), rolling circle replication (RCR) and conspicuous multimeric circular ssDNA (cms). In addition, 2.3-kb viral DNA fragment would be replicated by a single fork traversing the fragment (Y) and by two converging forks (Y) that met at the termination site for DNA replication.
Episomal Replication of BPV-1 DNA in Long-Term Virion-Infected S. cerevisiae Cultures
DNA Replication Intermediates in Long-Term BPV-1-Infected S. cerevisiae Cultures
Diagrammatic Explanation of BPV-1 DNA Replication Intermediates in S. cerevisiae
-
It has been established that PVs, the small double-stranded DNA viruses, infect epithelial tissue of their mammalian hosts and replicate in mitotically active basal keratinocytes in which the viral genomes are maintained as a nuclear plasmid (episome). Generally, viruses causing acute infections usually transform the infected cell into a virus factory, eventually leading to cell death and release of the viral progeny (Lipsitch and O'Hagan 2007). However, PVs do not kill the infected cell. Instead, PV infections persist mostly for decades but are not very productive (Cubie 2013). The PVs in persistently infected mammalian host epithelia rely upon the host replication machinery for their replications. However, it still lacks mammalian cell systems and an animal model for the vegetative replication of HPVs. We previously demonstrated that replication of BPV-1 DNA was present in virion-infected S. cerevisiae cells. Here we report further that episomal replication of BPV-1 DNA persistently present in virions-infected S. cerevisiae by long-term culture (up to 108 days). The persistent replication of BPV-1 DNA in S. cerevisiae is attributed to several physiological factors that DNA replication machinery is generally conserved between humans and S. cerevisiae (Chattopadhyay et al. 2005) and the cellular origin recognition (ORC) complex is functionally conserved from yeast to mammalian cells (Vashee et al. 2001). Also, PVs possess autonomously replicating sequence-like elements that control viral DNA replication (Rogers et al. 2008). The consensus ARS sequence within the BPV-1 genome shares with yeast (Chuang and Kelly 1999).
In our experiments, we extracted the episomal BPV DNA from the long-term virion-infected yeast cultures (up to 108 days), proved the episomal replication of BPV-1 DNA in yeast cells by 1-DAGE and DNA Southern blot hybridization. It was interested to note that the episomal replication of the BPV-1 DNA could be divided into three stages, active replication, weak replication and stable replication over the three-time courses. The active replication of the BPV-1 DNA at the early stage (up to 16 or 23 days) could be that the replicating molecules had a high degree of superspiralization in at least part of the replicating genome, due to the intensive ongoing elongation of the synthesized DNA strand. The weak replication of the BPV-1 DNA in the middle stage (day 23–34 or 44) may be associated with that the replication of BPV-1 DNA could produce dramatic biochemical and structural changes, which might cause yeast cell damage. That may explain that the viral RNA transcription was very weak (Chen et al. 2021) and the virion-infected yeast cells grew slowly and unhealthy. At the late stage (day 45–108), the stable replication might be ascribed to the viral latency indicating that the BPV-1 virus was going to lie dormant within yeast cells, which was a typical characteristic shared by PVs (Araldi et al. 2017).
A previous study has reported that replication of BPV-1 genome in a subclone of 1ID13 mouse fibroblasts latently transformed with BPV-1 DNA occurs as a mixture of extrachromosomal circular monomers and oligomers (Schvartzman et al. 1990). However, no study has been reported to use real virions isolated from bovine papilloma to infect S. cerevisiae and mammalian cells and investigate the replication patterns and intermediates of the viral DNA. Previously, we observed that BPV-1 DNA partially digested with HindIII in the short-term BPV-1 virion-infected S. cerevisiae culture showed a single replication bubble, a typical replication pattern (Zhao and Frazer 2002a). In the present study, we investigated the replication patterns of BPV-1 DNA in BPV-1 infected S. cerevisiae culture over a long time period (82 days) by means of 2-DAGE and Southern blot hybridization. It has been reported that analysis of replication intermediates in HPV-18 might help to elucidate the complicated replication mechanisms of its viral DNA replication (Orav et al. 2015). Indeed, multiple replication intermediates have been detected in the present study. The detected replication intermediates including linear forms, single-stranded, double-stranded and heterogeneous double-stranded DNAs appeared to be the autonomous viral DNA strands, which were steady over the time period. The occurrences of the linear monomers, dimers, trimers, and higher oligomers occurred in S. cerevisiae may be well explained by a published study in mammalian cells (Schvartzman et al. 1990). Initiation of DNA replication of BPV-1 occurred near the centre of the EcoRI-BamHI 5.6 kb fragment. The EcoRI-BamHI 2.3 kb fragment replicated as a DNA molecule containing a termination site for DNA replication and also by means of a single fork traversing the fragment from one end to other. Thus, multiple copies of BPV-1 DNA occurred at a single site in a head-to-tail tandem array, a partial digestion with a restriction enzyme that cut the BPV-1 genome only once would generate a series of linear monomers, dimers, trimers, and higher oligomers (Schvartzman et al. 1990). The other replication intermediates such as conspicuous multimeric circular ssDNA, rolling circle replication and recombinant-dependent replication intermediates occurred in S. cerevisiae are very complex, which appear to be associated with the utilization of templates and their length (Erdmann, 2010; Erdmann et al. 2010). The Y-shaped replication intermediates containing branched molecules are probably due to a delayed fashion with a beginning of leftward replication (Belanger et al. 1996). Thus, the multiple replication intermediates detected in the present study may be ascribed to that BPV-1 has different replication modes in S. cerevisiae. Two replication modes: an ordered once-per-S-phase fashion and a random fashion have been reported for the replication of BPV-1 DNA in eukaryotic cells (Berg et al. 1986; Gilbert and Cohen 1987; Nallaseth and DePamphilis 1994; Ravnan et al. 1992).
In viruses such as hepatitis B virus (HBV) and hepadnaviruses, higher oligomer cccDNA is a key replication intermediate in the viral life cycle (Cheng et al. 2020; D'Arienzo et al. 2021; Li et al. 2017; Lucifora et al. 2017). The cccDNA persisted in the nucleus is a long-lived nucleosome-associated minichromosome, which is the main nuclear transcription template for producing all viral RNAs (Hong et al. 2017; Luo et al. 2020). Recently, covalently closed circular RNAs (cccRNAs) have been described in the human DNA tumor viruses Epstein-Barr virus (EBV) and Kaposi's sarcoma-associated herpesvirus (KSHV) (Toptan et al. 2018). In the present study, cccDNA was detected in the BPV-1 virion-infected S. cerevisiae cultures. However, the frequencies of cccDNA at the first stages (day 3–16) were significantly higher than those at the third stage (day 45–82) while the cccDNA appeared to be suppressed at the second stage (day 23–34) due to the unhealth growth of the virion-infected yeast cells. The high frequency of cccDNA at the first stage might directly contribute the high transcription of BPV-1 RNA (Chen et al. 2021). In addition, each time point at the third stage (day 45–82) that showed to have only one cccDNA occurred might suggest that replication of BPV-1 viral DNA was conserved to maintain its genomic DNA at low-copy numbers for the latent infection.
Recently, Toots and colleagues reported that several novel HR-HPV-specific inhibitors were identified to inhibit the HPV replication in the cells (Toots et al. 2017). These HR-HPV-specific inhibitors target Tdp1 and PARP1 leading to blocking HPV genome replication, colliding replication forks and eventually producing aberrant DNA replication intermediates (Toots et al. 2017). Thus, whether the identified HR-HPV-specific inhibitors could target BPV-1 L2 proteins, specifically the L2 CPP to prevent BPV genome replication leading to forming aberrant DNA replication intermediates is worthy of future study in BPV-1 virion-infected S. cerevisiae.
In conclusion, we have shown three episomal replication patterns of BPV-1 genomic DNA in virion-infected S. cerevisiae for long-term cultures (up to 108 days) by means of DAGE and Southern blot hybridization. BPV-1 genome could replicate as extrachromosomal dsDNA circular plasmids and maintain stable copy numbers at the late stage in the virion-infected yeast cells. Our data have revealed that multiple replication intermediates detected include the linear monomers, dimers and trimers, higher oligomers especially cccDNAs, single-stranded DNA, double-stranded DNA, heterogeneous double-stranded DNA, multimeric circular ssDNA, rolling circle replication and recombinant-dependent replication intermediates. The occurrence of these viral replication intermediates will improve our understanding of BPV-1 genomic DNA replication in S. cerevisiae system.
-
This work was funded in part by grants from the National Nature Science Foundation of China (81772791 and 81172463).
-
KNZ conceived, designed, and supervised the study, and reviewed and revised the manuscript. QT, QL, WF, ZC, YZ and JC performed the experiments. PJ analysed data and drafted the manuscript. XX contributed to design and reviewed the data and manuscript. LZ supervised the study and reviewed the data and manuscript.
-
The authors declare that they have no conflict of interest.
-
This article does not contain any studies with human or animal subjects performed.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.














 DownLoad:
DownLoad: